Your consumer goods news from the world
Provided by AGPMultiple Myeloma Treatment Market Set for Robust Growth, Reaching US$ 14.6 Bn by 2033 | Persistence Market Research
The multiple myeloma treatment market is expanding rapidly, driven by innovative therapies, personalized medicine, and growing global healthcare access.
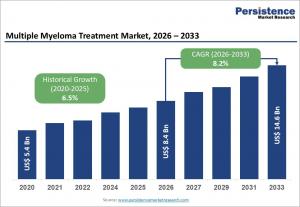
LONDON, UNITED KINGDOM, May 5, 2026 /EINPresswire.com/ -- The global multiple myeloma treatment market is poised for significant expansion, with the market size expected to reach US$ 8.4 billion in 2026 and projected to grow to US$ 14.6 billion by 2033, registering a compound annual growth rate (CAGR) of 8.2% during the forecast period. This growth is largely driven by the rising prevalence of multiple myeloma among aging populations, particularly in developed regions where life expectancy is increasing. The rising demand for innovative therapies has also spurred investments in drug development, further stimulating market expansion.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/13954
Key Industry Highlights
Targeted therapy is projected to account for the largest revenue share of nearly 30% in 2026, reflecting its clinical efficacy and widespread hospital adoption. Immunotherapy is expected to be the fastest-growing treatment type between 2026 and 2033, driven by the adoption of CAR-T therapies and monoclonal antibodies. North America is anticipated to dominate the market with an estimated 38% share in 2026, supported by advanced healthcare infrastructure, while the Asia Pacific region is forecasted to be the fastest-growing regional market due to rising private healthcare investment and digital health adoption. The market remains moderately consolidated, with multinational pharmaceutical companies leveraging strong R&D capabilities, strategic partnerships, and differentiated product portfolios.
Market Drivers
The increasing number of multiple myeloma cases in aging populations is a primary growth driver. In the United States, new cases are projected to reach 36,110 in 2025, predominantly affecting individuals aged 65 and above. Older patients typically present with comorbidities and slower immune responses, increasing complication rates and care requirements. This demographic trend has heightened demand for specialized oncology care, long-term disease management, and novel therapies, influencing clinical research funding and innovation.
Investment in new drug development also supports market growth. Public agencies, including the U.S. National Cancer Institute, allocated over US$ 7.2 billion in FY 2025 to cancer research, including multiple myeloma studies. Government-backed research initiatives reduce barriers for pharmaceutical companies, accelerate clinical trials, and foster the development of next-generation therapies. This investment landscape encourages collaborations between academia and industry, strengthening the pipeline of innovative treatments.
Market Challenges
Access to advanced therapies is constrained by limited specialized treatment centers. Complex therapies such as CAR-T cell treatments and stem cell transplants require multidisciplinary teams, advanced infrastructure, and rigorous regulatory compliance. Patients in rural or underdeveloped areas often face logistical challenges, delaying treatment initiation and affecting adherence. Establishing new centers demands significant capital, skilled personnel, and extended training programs, restricting expansion into underserved regions.
The high cost of advanced therapies further limits market growth. CAR-T therapies, monoclonal antibodies, and bispecific antibodies require extensive research, specialized manufacturing, and complex logistics, contributing to elevated treatment prices. Variations in reimbursement policies and limited competition in certain therapy segments create affordability gaps, affecting patient access and adoption. Operational and infrastructure requirements amplify cost pressures, influencing treatment protocols and overall market dynamics.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/13954
Opportunities
Targeted and personalized therapies offer significant potential due to their precision in attacking cancer cells while minimizing harm to healthy tissues. Monoclonal antibodies targeting CD38 and bispecific antibodies have demonstrated improved progression-free survival and treatment outcomes. Digital tools for monitoring and diagnosis are increasingly integrated into patient care, enabling timely clinical decisions, symptom tracking, and real-time access to health records, enhancing treatment adherence and monitoring.
Regional Insights
North America leads the market, driven by advanced healthcare systems, early adoption of CAR-T and bispecific therapies, and strong insurance coverage. Europe maintains a prominent position due to clinical research networks, reimbursement frameworks, and the widespread use of targeted therapies and CAR-T treatments. Asia Pacific is the fastest-growing market, supported by rising healthcare investment, digital health adoption, and expanding oncology infrastructure in China, India, Japan, and South Korea.
Market Segmentation
By Treatment Type
Chemotherapy
Immunotherapy
Targeted Therapy
Stem Cell Transplant
Radiation Therapy
By Stage of Disease
Stage I
Stage II
Stage III
By Drug Class
Immunomodulatory Drugs
Proteasome Inhibitors
Monoclonal Antibodies
CAR-T Cell Therapy
Bispecific Antibodies
Corticosteroids & Chemotherapy Agents
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East & Africa
Checkout Now & Download Complete Market Report: https://www.persistencemarketresearch.com/checkout/13954
Competitive Landscape
The global multiple myeloma treatment market is moderately consolidated, dominated by companies such as Bristol-Myers Squibb, Takeda Pharmaceutical Company, Johnson & Johnson, Amgen, AbbVie, Celgene, Pfizer, Sanofi, Gilead Sciences, Regeneron Pharmaceuticals, Roche, Karyopharm Therapeutics, BeiGene, and Legend Biotech. Competitive strategies focus on product differentiation, clinical efficacy, strategic partnerships, and global distribution. Mid-sized biotech firms contribute through niche innovations and collaborations with larger players.
Recent Developments
In early 2026, research at Emory University highlighted advancements in quadruple frontline regimens and minimal residual disease testing. Technical University of Munich demonstrated reactivation of CAR-T therapies using proteasome inhibitors, while Memorial Sloan Kettering Cancer Center reported improved survival through novel drug combinations and FDA-approved monoclonal antibody regimens. These developments underscore the ongoing evolution of treatment paradigms, extending patient survival and improving quality of care.
Conclusion
The multiple myeloma treatment market is undergoing dynamic growth, fueled by rising disease prevalence, technological innovation, and increasing investment in targeted and immunotherapy solutions. While challenges such as high treatment costs and limited specialized centers persist, advancements in personalized medicine, digital monitoring tools, and regional healthcare expansion are set to drive sustained market growth through 2033.
Read Related Reports:
Microtainer Blood Collection Market: Microtainer blood collection market grows from US$1.7 Billion in 2026 to US$2.8 Billion by 2033 at 5.7% CAGR, driven by rising global diagnostic testing.
EPRO, E-patient Diaries and eCOA Market: The global ePRO, ePatient Diaries and eCOA market will rise from $2.9B in 2026 to $7.9B by 2033, posting a strong 15.3% CAGR globally and steadily growing
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.